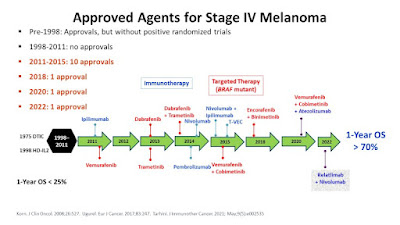
Originally posted in 2017, sadly, this primer has needed little in the way of updates since. Still, there have been a few FDA approvals, meds I didn't include in the first rendition, and some news in the research so it is getting a reboot. Initially created to save re-writes for those in need, I still answer melanoma questions on boards or via email at least every other week, but want to emphasize that this is not an all inclusive listing. Rather, this is a basic guide to use in starting your research or discussions with your provider regarding melanoma care. As recently, as 2010, NONE of the current, most effective treatments for melanoma were FDA approved. Since then, doctors have become more knowledgeable about consistently offering and better skilled in managing these treatments. Still, it is essential that you be seen by an oncologist who specializes in, or at the very least, has treated many patients with melanoma. Sometimes a picture is worth a thousand words ~
Here we go:
SURGERY
Surgery remains a good choice for many melanoma patients. Clearly this is the case for a new cutaneous lesion. Surgery results in an immediate decrease in your tumor burden - almost always a good thing. However, with data showing good results in NEO-adjuvant treatment, the possibility of using intralesional therapy, or if you are looking for a clinical trial, there are times when measurable disease is needed, so a discussion of these things before surgery is important. However, for patients with advanced disease decreasing tumor burden through surgery remains an important option for increased survival. This 2019 report addresses some of the conundrum: Cut it out!!! Prolonged overall survival following metastasectomy in Stage IV melanoma
RADIATION
Radiation, when combined with immunotherapy or targeted therapy, can be a very good treatment option for melanoma. Together, radiation and systemic therapy can illicit responses that are greater than either treatment used as a single agent. However, targeted radiation (SRS - stereotactic radiation or Gamma Knife) is the most effective whether you are talking about brain tumors or lesions in the body. We have learned that whole brain radiation (WBR) is not the most effective way to treat melanoma and can lead to debilitation. While there are those who must avail themselves of this treatment due to extreme circumstances, it should not be the first recommendation right out of the box for those with brain tumors. Even multiple brain mets can be treated simultaneously with SRS. Here are zillions of reports regarding the effectiveness of using radiation WITH immunotherapy: Radiation WITH immunotherapy Here is a report from 2019 regarding the use of radiation prior to targeted therapy: Better melanoma results with radiation BEFORE BRAFi (at least in this report)
IMMUNOTHERAPY
These are treatments that push our immune systems into action. Side effects (as you might imagine) are usually related to an 'over activation' of the immune system. Common side effects include - fatigue, rashes, joint pain. More complicated side effects are inflammation of the lungs (pneumonitis) and colon (colitis) with difficulty breathing and wheeze or diarrhea and abdominal discomfort, respectively. Patients can experience problems with thyroid function and other glands of the endocrine system. Responses take time. Experts are known to advise other docs to be 'patient with the patient!' Immunotherapy works best with the lowest tumor burden.
Response rate and side effects for advanced melanoma patients:
Both anti-PD-1 drugs as single agents effect about a 40% response rate in melanoma. They can work in the brain and the body. Median time to response is about 3 months. But, there are outliers, with documented responses, that do not occur until 6 - 9 months. Here's a cool graph...
 |
| Here's a post with more info: Time to Response...Ipi vs Nivo and ipi |
Side effects are similar for both drugs and are those typical for immunotherapy, but less severe than those encountered with ipi. As expected, the ipi/nivo combo has greater side effects than when nivo or pembro are used alone. On the topic of side effects...they SHOULD be treated!!! As quickly as possible. At times, a break from medication and immunosuppressive drugs are required. While oncologists not familiar with immunotherapy may fear decreased therapeutic response if steroids are used...the preponderance of the data indicates that THIS IS NOT THE CASE!!!! Clearly, one should not take immunosuppresive drugs unless absolutely needed. Many patients require varying doses of steroids in order to tolerate necessary, life saving melanoma treatments and go on to do well! Further, folks with pre-existing autoimmune disease can be managed on immunotherapy and gain a response as well. Here are a zillion reports on all of that jazz: What to do about immunotherapy if you need steroids or have a pre-existing autoimmune disease?
When Pembro is used as a single agent = is dosed at 2mg/kg with max of 200 mg IV every 3 weeks - for one year as adjuvant, end point undefined for advanced melanoma patient. Nivo as single agent = is dosed at 240 mg IV every two weeks or 480mg IV every 4 weeks - for one year as adjuvant, endpoints vary for advanced melanoma patients. When ipi is combined with nivo, response rates in melanoma rise to 50+%, though side effects increase as well - mostly due to ipi. For the combo, dosage is: nivo at 1 mg/kg followed by ipi at 3 mg/kg on the same day, every 3 weeks for 4 doses, then nivo alone at 240 mg q 2 wks or 480 mg q 4 wks. endpoint varies. Many patients cannot tolerate all 4 doses of the ipi/nivo combo due to side effects. However, outcomes can be good even if you have to stop early. Here's a report from ASCO 2016: ASCO 2016 - Nivo plus ipi, CheckMate 069 trial....18 month OS similar even if you stop meds due to side effects!!! Further, the ASCO 2021 data (link above) notes "Clinical benefit response (CBR) after 1 or 2 doses of I/N may be predictive of long-term survival in advanced stage melanoma. Patients who have CBR after 1 or 2 doses of I/N may achieve a similar survival benefit with fewer doses of I/N." Finally, most folks who cannot tolerate the combo can go on to tolerate nivo alone, once their side effects are brought under control with a medication break and/or steroids.
TARGETED THERAPY
At this point in melanoma, the only approved targeted therapy is for patients whose tumor is positive for the BRAF V600 mutation. About 50% of melanomas are. However, researchers are looking at drugs that could target other points in the molecular pathway of melanoma. This diagram shows what I mean by "pathway"...
 |
| A Melanoma Molecular Disease Model (See the link below for credit and more info) |
Here's just one example from March of this year: What tangled 'paths' we weave: Nilotinib for KIT mutated melanoma and Buparlisib for the PI3K pathway in melanoma brain mets
But....for current purposes....I am focusing on the BRAF mutation. Here's a post I made a bit ago that really breaks down what BRAF is, what it means in melanoma, and how the drugs work: BRAF inhibitors for melanoma: Dabrafenib, Vemurafenib, Dabrafenib/trametinib combo. Answers!!!!!
Usually when we combine drugs, we end up with increased side effects. However, in the case of BRAF targeted therapy we now know that BRAF inhibitors should ALWAYS be given with a MEK inhibitor. Strangely enough, when the combo is given, patients experience better response rates, DECREASED side effects, and DECREASED rates of tumor work-around. The only exception is when MEK inhibitors are used as a single agent in specially mutated patients.
DRUGS, administration, and side effects:
BRAF inhibitor (BRAFi) drugs include: Vemurafenib (Zelboraf), Dabrafenib (Tafinlar), Sorafenib (Nexavar), and Encorafenib (Braftovi)
MEK inhibitors (MEKi) include: Trametinib (Mekinist), Cobimetinib (Cotellic) and Binimetinib (Mektovi)
These drugs are administered orally. So that's super cool. Dosing depends on the particular drug.
Side effects include joint pain, rashes, extreme sun sensitivity, development of benign skin cancers, fevers and sometimes liver toxicity.
EFFECTIVENESS and tumor work-around:
For patients who are BRAF positive, BRAF inhibitors combined with a MEK inhibitor have impressive response rates, clearing tumors rapidly, and often completely, in about 70-80% of patients and are effective in the brain and body. However, those responses are not very durable, with most tumors learning to work around the inhibition in about 7-9 months. BUT!!!! By using an "alternate dosing schedule" (one that is varied, rather than absolute with an 'every so many hours daily' dosing pattern), combining BRAFi with MEKi, as well as the development of the newer drugs that time can be stretched out a bit. Furthermore, despite the statistics, there are some melanoma peeps whose melanoma has been successfully managed for years on BRAF/MEK combo's!! Finally, some melanoma specialists use BRAF/MEK combo's in BRAF positive patients, to rapidly decrease the tumor burden, then switch the patient to slower acting, but more durable immunotherapy. Picking which targeted therapy to use can be difficult. Here are two posts that attempt to pull response rates and PFS out of the data ~
INTRALSIONAL (also referred to as 'intratumoral') THERAPY
Intralesional drugs include (but are not limited to):
T-VEC - also called OncoVEX, Imlygic, or Talimogene Laherparepvec - uses the herpes virus with GM-CSF and is the only intralesional currently FDA approved (2015) However, the following (and others) have been used in clinical trials:
CAVATAK - derived from the Coxsackievirus
T-VEC - also called OncoVEX, Imlygic, or Talimogene Laherparepvec - uses the herpes virus with GM-CSF
PV-10 - derived from Rose Bengal
HF10 - also derived from HSV
SD101 - a TLR9 agonist
IL-2 - see note above, is also being used
These drugs are injected directly into a relatively superficial melanoma tumor. They have been found to be effective in not only eradicating the tumor into which they have been injected, but 'by-stander' lesions as well. Researchers feel that they have the most promise when they are combined with a systemic treatment like immunotherapy. I summarized response rates, side effects, and pretty much everything else current about these drugs in these posts which include many links within them:
-------------------------------------------
I hope this primer will continue to be a helpful jumping off point for those in need. What has served me best in attaining effective treatment for my melanoma has been seeking out a melanoma specialist (or at least an oncologist who cares for many melanoma patients) and never being afraid to ask questions. Asking this question of my doctor may have been the most beneficial: "What treatment would you recommend if it were YOU or your brother, sister, wife, father, mother.... in need?"
I wish you all my very best. Hang in there. And as ever, with enduring thanks to the ratties! - love, c
P.S. If all the acronyms are driving you crazy, here's a post that defines at least some of them: Melanoma abbreviations ~ and random thoughts on posting melanoma crap-ola....
P.S.S. A sense of humor really does help!! AND FINALLY - while not all inclusive, this post from 2019 includes a list of world class melanoma specialists: Internationally renowned melanoma specialists: - c

No comments:
Post a Comment